Physical properties of two-component gas: a phase diagram of pure CO2,... | Download Scientific Diagram

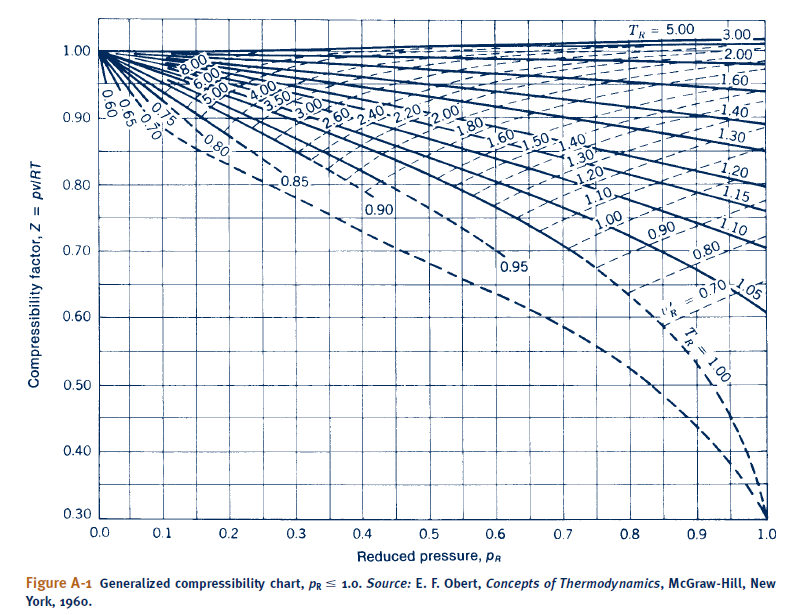
59.) One mole of CO2 occupying 0.4 litre 300 K and 40 atm pressure is (a) compressible (b) compressible (c) ideal (d) does not have a definite compressibility. 2018 (JIPMER)

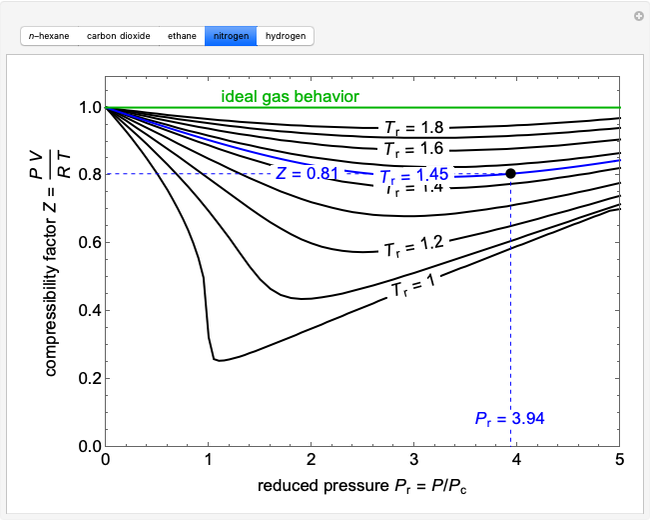
physical chemistry - Compressibility Factor Graph - Which gas attains a deeper minimum? - Chemistry Stack Exchange
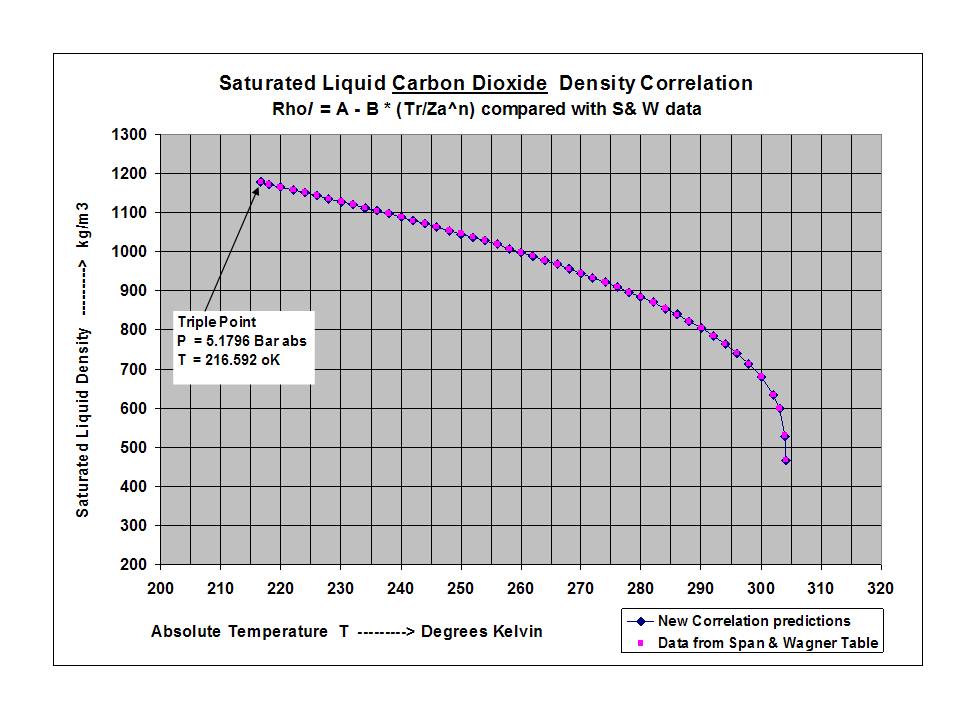
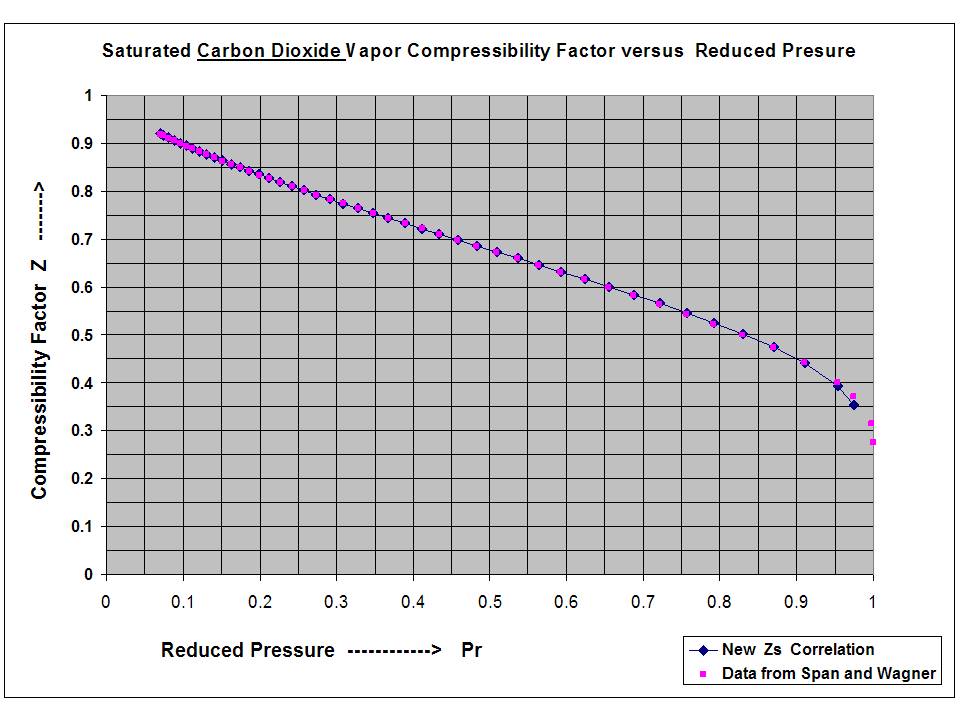
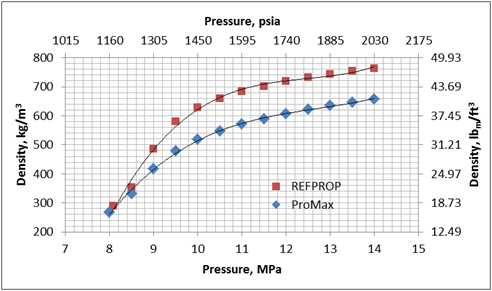
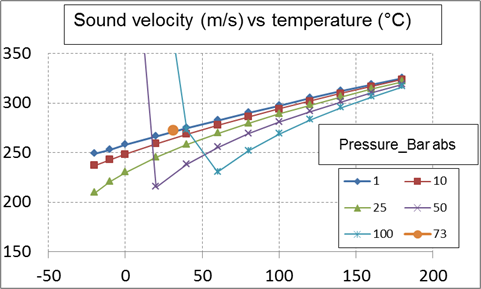
New compact Equations for the Compressibility Factor Z and Density of Liquid and Vapor Carbon Dioxide | Chem-Eng-Musings

Figure 3 from Compressibility Factor of Gas with High Content of CO2 in Changshen Gas Reservoir | Semantic Scholar

Carbon dioxide compressibility factor determination using a robust intelligent method - ScienceDirect

physical chemistry - Why do some gases have lower value of Z for a particular pressure? - Chemistry Stack Exchange
Compressibility of supercritical CO2 showing that the supercritical... | Download Scientific Diagram

Carbon dioxide compressibility factor determination using a robust intelligent method - ScienceDirect

Figure 2 from Compressibility Factor of Gas with High Content of CO2 in Changshen Gas Reservoir | Semantic Scholar

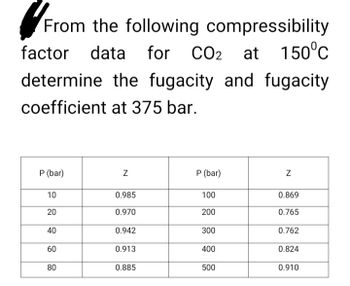
Compressibility factor for CO2 at 400 K and 71.0 bar is 0.8697. Molar volume of CO2 under these conditions is:

![PDF] Measurement of Z-factors for carbon dioxide sequestration | Semantic Scholar PDF] Measurement of Z-factors for carbon dioxide sequestration | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/68e746895d9735e5ff297e3058fa1b33defaefd6/7-Figure6-1.png)